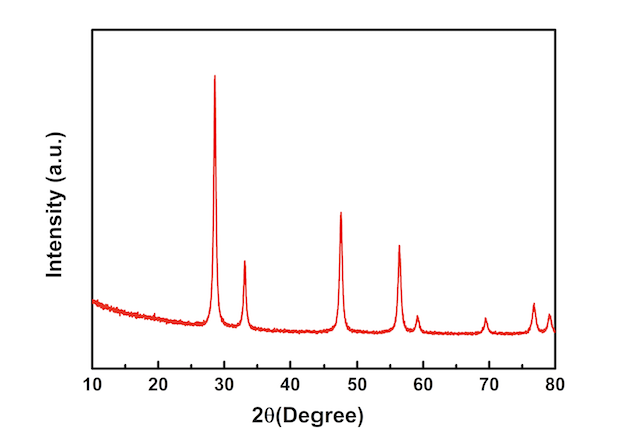
When an X-ray encounters an atom, its energy is absorbed by the electrons, and is then released in the form of a new X-ray in a phenomenon called “elastic scattering”. These crystals have a regular arrangement of atoms, each surrounded by a cloud of electrons. Most materials consist of many small crystals. The X-ray source and the detector move at different angles in a synchronized motion. In a typical XRD experiment, the sample is illuminated with a beam of X-rays. X-rays have wavelengths in the range of 10 −10 m, the same order of magnitude as the distance between atoms (measured in Ångströms, Å). Principles of XRD – Measuring the Distance Between Atoms 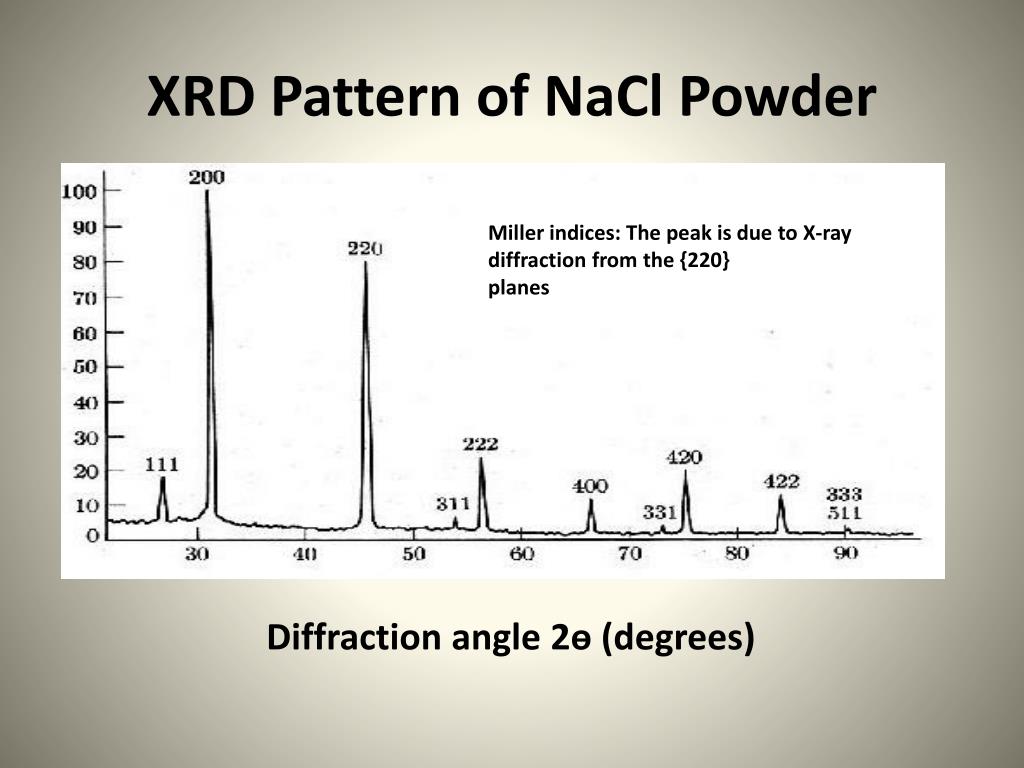
XRD was developed at the beginning of the 20 th century and its application to the analysis of crystals led to the award of the Nobel Prize in Physics to Sir William Henry and William Lawrence Bragg in 1915 – one of the many Nobel Prizes associated with XRD. XRD is a non-destructive technique and helps scientists to understand how atoms are arranged and how such arrangements can affect the behavior of materials or can be used to determine the structure of complex molecules. Such materials are normally crystalline (or partially crystalline) and can range from single crystals, thin films, and mixtures of powders. The X-ray diffraction (XRD) technique is used to characterize the structure of a wide variety of materials on the atomic and molecular levels.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
